Optimizing Laser Atherectomy for Different Lesion Morphologies
Journal of Critical Limb Ischemia, March 2021
Objective
To understand the impact of fluence and repetition rate on outcomes of superficial femoral artery (SFA) and popliteal artery laser atherectomy based on lesion type (calcific, homogenous, heterogeneous, and restenosis).
Methods
Patients with de novo or restenotic (≥50%) SFA and popliteal artery atherosclerotic disease were enrolled. All lesions were sequentially treated with Turbo-Power laser atherectomy (Spectranetics) at 3 predetermined intensity settings: low (fluency, 40 mJ/mm2; repetition rate, 60 Hz); medium (fluency, 60 mJ/mm2; repetition rate, 40 Hz); and high (fluency, 60 mJ/mm2; repetition rate, 60 Hz). Angiography and intravascular ultrasound (IVUS) were performed to characterize plaque morphology and evaluate residual stenosis. Follow-up was 30 days and medical records were reviewed through 12 months for adverse events.
Results
Forty-five patients with 57 lesions (12 homogenous, 15 heterogeneous, 15 calcific, and 15 restenotic) were enrolled. Rutherford classification ranged from 2-5, average lesion length was 98.2 ± 91.2 mm, and average diameter stenosis was 82.5 ± 17.9%. Compared with baseline, all lesion types had significant improvement in final postprocedure (atherectomy + any adjunctive therapies) diameter stenosis. Prior to adjunctive therapy, the heterogeneous and restenosis groups saw improvement in minimum lumen area following each stage of the laser treatment. However, the calcific and homogenous groups saw little change in minimum lumen area between the medium- and high-intensity laser treatments. Within 6 months, a total of 6 patients had target-lesion revascularizations. No major amputations or deaths occurred through follow-up.
Conclusion
Laser intensity settings during atherectomy should be selected based on lesion morphology. IVUS was essential in defining plaque morphology.
Introduction
Personalized medicine refers to a model of medical practice where diagnosis and subsequent treatment are customized to the individual patient. Recent technological advances have now allowed us to enter “the era of personalized care,”1 a practice which is advantageous due to diagnostic testing and therapies tailored to the individual patient, resulting in reduced treatment time and associated costs.2 This approach to care has been adopted in various medical fields, including oncology and immunology, and is quickly being adapted to the field of cardiovascular therapy.3
This is especially evident in the diagnosis and treatment of peripheral arterial disease (PAD), which utilizes multiple modalities for diagnosis and treatment. Patients with PAD have heterogeneous disease presentation and underlying pathology. Characterizing the individual patient’s disease with advanced imaging can hypothetically help tailor the interventions that would deliver the best outcome.
Of particular interest is the potential of atherectomy to treat lower-extremity PAD. Compared with more traditional endovascular therapies, the optimal device settings, indications, and treatment strategies are not well described for laser atherectomy. Additionally, the optimal device settings for different lesion morphologies are not well defined.
We aimed to discern the optimal laser treatment protocol for maximizing debulking in an array of PAD-related superficial femoral artery (SFA) and popliteal lesion morphologies. In doing so, we combine imaging and interventional therapies with the goal of personalizing and optimizing future treatment algorithms.
Methods
Study design
This was an observational, single-center, non-randomized trial that enrolled patients with atherosclerotic lesions of the SFA and above-the-knee popliteal artery. Angiography and intravascular ultrasound (IVUS) were used to categorize the patients into the following groups based on their lesion type: homogenous, heterogeneous, calcific, and restenotic. Homogenous lesions comprised soft plaque, heterogeneous lesions contained both soft plaque and intermittent calcific specks (<50%), calcific lesions contained primarily insoluble calcific compounds (>50%) with intermittent soft plaque, and restenotic lesions were those that were previously treated at the same location and suffered recurrent lumen loss. Lesion categorization was performed with IVUS by 1 experienced principal operator for all lesions.
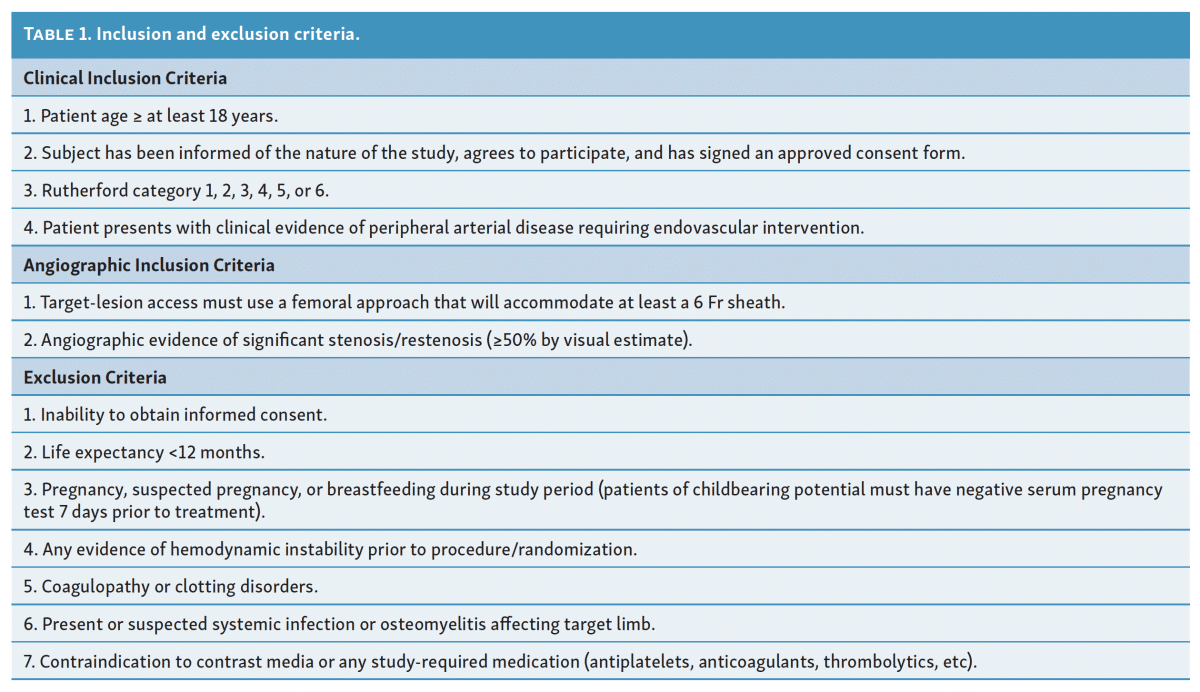
Patients with PAD of any Rutherford class were eligible to enroll in the study if they had de novo lesions requiring endovascular intervention, or angiographic evidence of ≥50% restenosis at a previously treated lesion. Full inclusion/exclusion criteria are listed in Table 1.
The study was conducted in accordance with the Declaration of Helsinki and was approved by the Western institutional review board. All participating patients were required to provide informed consent prior to enrollment.
Investigational device
The computer-controlled CVX-300 XeCl Excimer Laser unit (Spectranetics) was used during each procedure. Following initial calibration, the user could adjust the fluence and repetition rate throughout the procedure. A 2.0 or 2.3 mm Turbo-Power laser atherectomy catheter (Spectranetics) was used, which is compatible with a 6 Fr or 7 Fr sheath.
Procedure
Standard digital angiography performed in 3 views and IVUS were used at the start of the procedure to document lesion type, vessel diameter, and degree of stenosis. The Turbo-Power catheter was then passed through the vessel at 3 predetermined settings: low, medium, and high. Each laser setting corresponded to a specific combination of fluency (mJ/mm2) and repetition rates (Hertz). The low setting had a fluency of 40 mJ/mm2 with repetition rate of 60 Hz, the medium setting had a fluency of 60 mJ/mm2 with repetition rate of 40 Hz, and the high setting had a fluency of 60 mJ/mm2 and repetition rate of 60 Hz. The exact numerical values for fluency and repetition rate were determined by bench testing completed at Spectranetics.
When using the laser, the investigator ensured the laser catheter tip was in contact with the tissue and the laser was advanced at a speed of approximately 0.5-1 mm/s. A flush and infusion technique with normal saline solution was continuously utilized at the ablation site to clear the site of contrast media and blood, and to prevent microbubble formation and collapse. The infusion was administered in a constant fashion while the laser system was activated.
Following each pass of the atherectomy catheter, angiography and IVUS were used to assess vessel diameter. Once >50% improvement in diameter stenosis was achieved, treatment with laser atherectomy was ended. Subsequently, the investigator would determine if adjunctive therapy was necessary (such as percutaneous transluminal angioplasty with plain old balloon angioplasty or drug-coated balloon [DCB], or stenting). At the termination of the procedure (including all adjunctive therapies), angiography and IVUS were performed to discern the degree of residual stenosis at the target treatment site. IVUS and angiographic data were adjudicated by an independent core lab (Syntactx).
All patients were required to complete a 30-day follow-up, either in person or by phone call. Each patient’s medical records were reviewed through 12 months post procedure for all adverse events, including target-lesion revascularization (TLR) and/or target-vessel revascularization (TVR).
Endpoints
The primary endpoint was the final reduction in target-lesion stenosis post procedure, as determined by angiogram and IVUS. Secondary endpoints included residual stenosis after each laser pass, posttreatment luminal quality relative to preintervention (as determined by IVUS), procedural complications, presence of flow-limiting dissection post procedure, freedom from TLR through 12 months, freedom from TVR through 12 months, and incidence of major adverse events (MAEs). MAE was defined as all-cause death, major amputation in the target limb, or TLR (surgical or interventional) from procedure to 30 days (± 7 days). Acute procedural success was defined as investigator-determined residual stenosis of ≤30% in the target lesion following laser treatment but prior to any adjunctive or bail-out procedures.
Statistical analysis
Image and data analyses were conducted by a core laboratory (Syntactx). Descriptive statistics were used to present data from the study. Continuous variables are presented as mean ± standard deviation, whereas categorical variables are presented as fractions (percentage). Student’s t-tests were used to compare means; paired t-test were used to compare baseline and final measurements.
Results
Patient characteristics
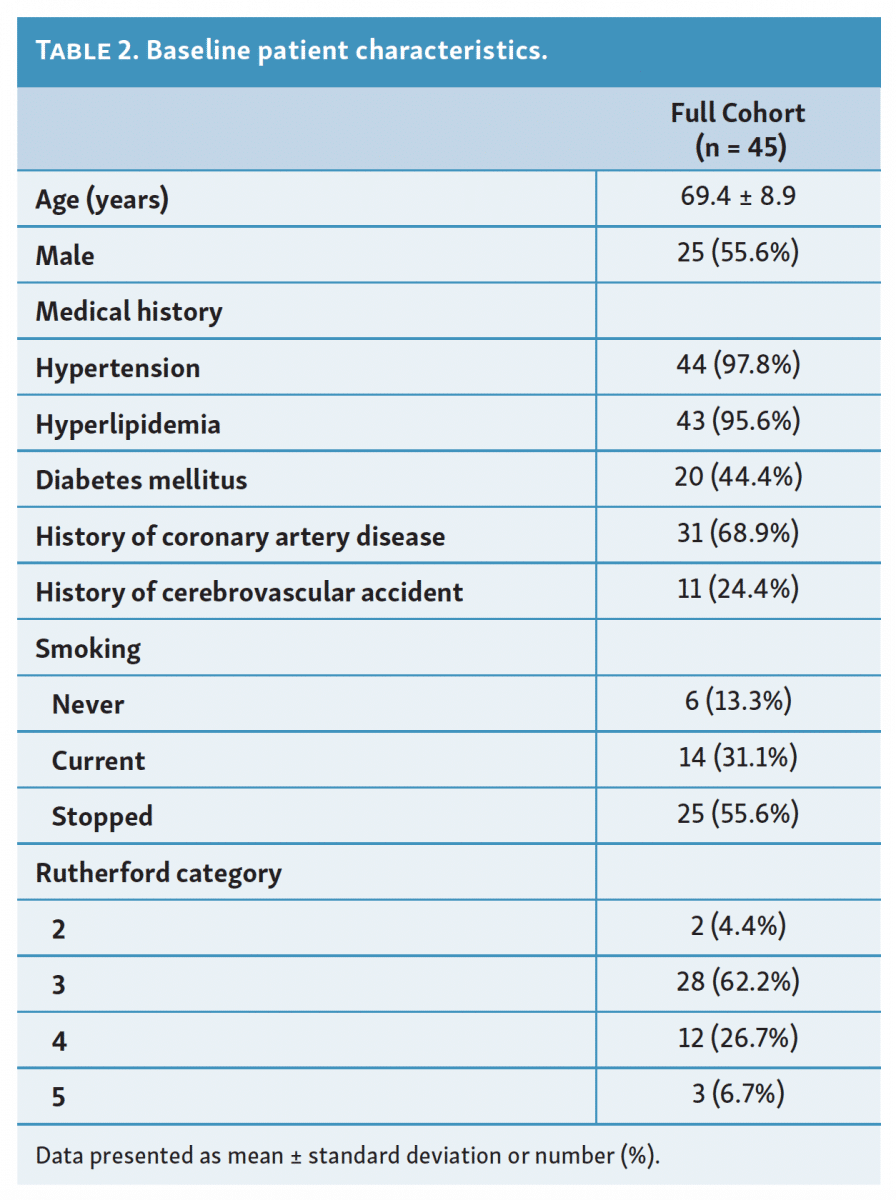
This observational, single-center, non-randomized trial enrolled 45 patients with 57 lesions (mean age, 69.4 ± 8.9 years; 25 men [55.6%]) with atherosclerotic lesions of the SFA and above-the-knee popliteal artery. The most common comorbid conditions in the study population were hypertension in 44/45 patients (97.8%) and hyperlipidemia in 43/45 patients (95.6%). The remaining baseline clinical characteristics are summarized in Table 2. Most patients had a history of either former (55.6%) or current tobacco use (31.1%).
While lifestyle-limiting claudication (Rutherford category 3) was the most common clinical presentation of PAD in the study population (in 28/45 patients; 62.2%), one-third of enrolled patients presented with critical limb ischemia (CLI; Rutherford 4 and 5).
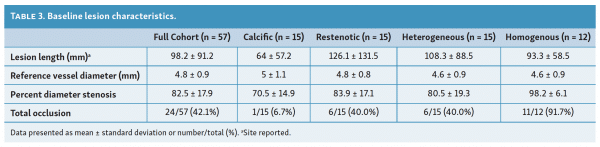
Mean lesion length was 98.2 ± 91.2 mm (range, 10-420 mm)and average percent diameter stenosis was 82.5 ± 17.9% (Table 3). Chronic total occlusions were present in 24/57 lesions (42.1%). Some degree of calcification was present in 36/57 lesions (63.2%).
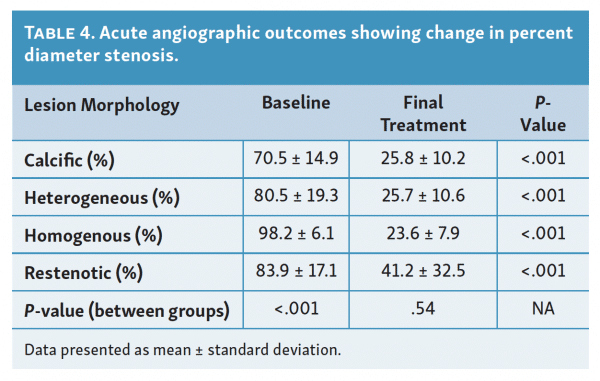
Baseline angiographic measurements showed that the group with homogenous-type lesions started with the highest percent diameter stenosis (98.2 ± 6.1%) (Table 4). Conversely, the calcific lesion group started with the lowest percent diameter stenosis (70.5 ± 14.9%).
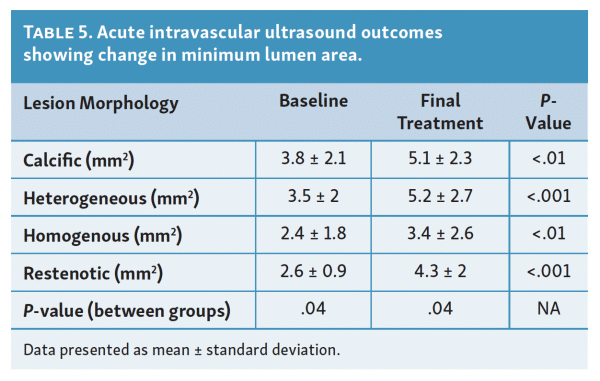
Utilizing IVUS for baseline measurements, the average minimum luminal area (MLA) was 3.1 ± 1.8 mm2. As would be expected based on the above angiographic findings, the average MLA was largest in the calcific group (3.8 ± 2.1 mm2) and smallest in the homogenous group (2.4 ± 1.8 mm2) (Table 5).
Procedure characteristics
Procedural success rate was 78.6% (45/57 lesions). Every patient in the study ultimately received adjunctive treatment, the most common of which was angioplasty with DCB in 50/57 lesions (87.7%). DCB along with adjunctive PTA was the only adjunctive therapy in 31/57 lesions (54.4%), and was used in combination with stent placement in 10/57 lesions (17.5%) or with thrombectomy in 2/57 lesions (3.5%). There were 5 lesions treated with DCB alone and 2 lesions treated with DCB plus stent placement. The remaining 7 patients were treated with PTA alone (3/57 lesions; 52.6%) or along with other adjunctive therapies, such as stenting and/or thrombectomy (4/57 lesions; 7.0%). Distal protection devices were used in 21/57 lesions (36.8%).
Primary and secondary endpoints
All lesion types experienced statistically significant improvements in degree of stenosis following final treatment (Table 4). The greatest change in percent diameter stenosis compared with baseline was seen in the homogenous group (98.2% vs 23.6% after final treatment; P<.001). The smallest change was seen in the restenosis group (83.9% vs 41.2% after final treatment; P<.001).
In addition to baseline, IVUS was utilized to measure MLA after each pass with laser atherectomy, but prior to any adjunctive therapy. The baseline MLA, MLA following final pass of the atherectomy device, and the change between the two values are summarized in Table 5. Although all lesion types experienced a statistically significant increase in MLA after final atherectomy treatment, the average change was greatest for the heterogeneous and restenotic groups (1.8 mm2).
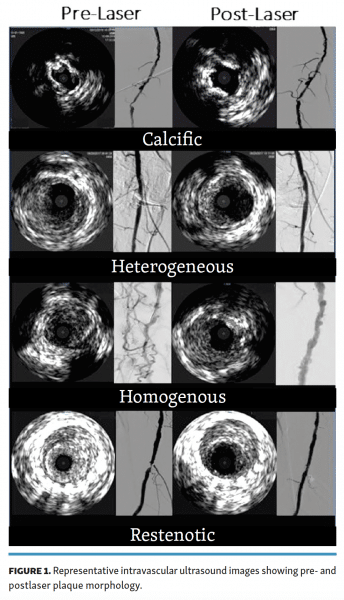
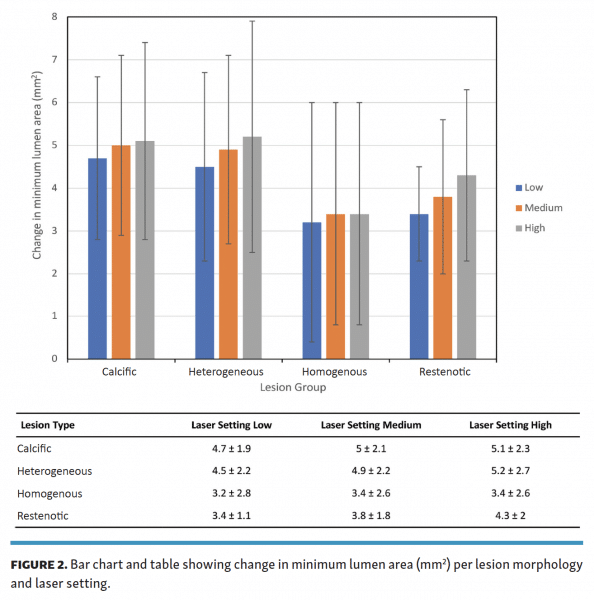
The changes in MLA following treatment with each laser setting are stratified by lesion type in Figure 1. After each pass with the laser, the calcific, heterogeneous, and restenotic lesions all saw a step-wise increase in mean MLA of 0.2 mm2, 0.4 mm2, and 0.5 mm2, respectively. However, this predictable stepwise luminal gain was not demonstrated on treatment of the homogenous group (Figure 2). While homogenous lesions saw a mean MLA gain of 0.2 mm2 after treatment transitioned from the low to medium setting, there was no appreciable interval increase after a third pass with the high setting.
Reinterventions
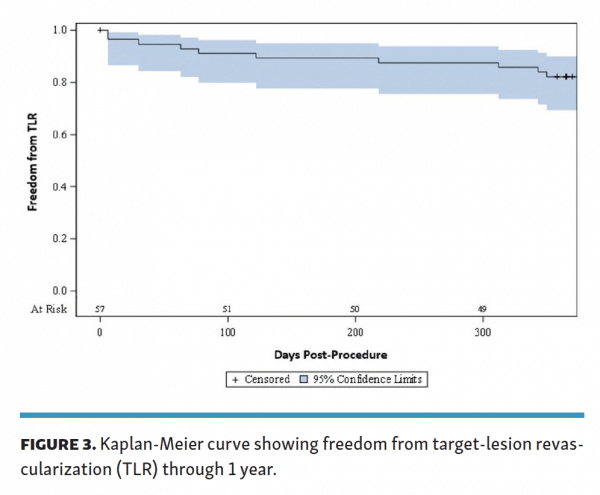
The rate of freedom from TLR in the entire cohort at 6 months (180 days) was 89.5%. Following the index procedure, a total of 6 lesions required TLR within 6 months and 10 lesions required TLR within 12 months (Figure 3). Four of the 10 lesions (40.0%) were from the calcific cohort and 6/10 lesions (60.0%) were from the restenotic cohort. These occurred more frequently in older patients (mean age 78.7 ± 4.2 years vs 68.8 ± 9.3 years). Only 6 of the 10 lesions (60.0%) requiring TLR within 12 months post procedure initially met the definition of procedural success. The rate of procedural success was 82% in the remainder of the study group.
The mean lesion length for the 6 patients who underwent TLR during the follow-up period was 176.7 ± 158.8 mm compared with 88.9 ± 77.3 mm in those who did not experience TLR. Calcification levels, percent diameter stenosis, and total occlusion rates in these 6 patients were largely comparable to the remaining study participants. TVR was noted in 13 patients within 12 months following the index procedure. There were no instances of major amputations or documented deaths during the follow-up period; however, the study population included few patients with Rutherford class 5 disease.
Discussion
Excimer lasers have been widely used to treat lesions containing plaque, thrombus, neointimal hyperplasia, and calcium.4 They were first used in the coronary arteries,5 but eventually received approval for use in the peripheral vasculature. They contain optical fibers located in the catheter tip that transmit pulses of light in the ultraviolet spectrum with short wavelengths (308 nm) and high energy. This energy ablates hyperplastic tissue in atherosclerotic plaques, thereby improving diameter stenosis and debulking in-stent restenosis.6 Unlike other atherectomy tools, excimer lasers allow for controlled, sequential, and directional ablation, as evidenced in the CELLO (CliRpath Excimer Laser System to Enlarge Lumen Openings) study. This leads to minimal thermal injury and decreases the risk of distal embolization. In fact, less than half the patients in this pilot study required distal protection and there was only 1 recorded instance of embolization thought to be related to the study device.4
Results from the EXCITE trial also illuminated the capacity of laser atherectomy to perform as a vessel preparation tool in the setting of in-stent restenosis. The patients who were treated with excimer-laser ablation plus adjunctive PTA fared significantly better during short- and mid-term follow-up than patients who only received PTA.6 Three years later, Kokkinidis et al published results from their study comparing patients treated with laser atherectomy + DCB with patients treated with laser atherectomy + PTA.7 Patients in the laser + DCB group had significantly higher rates of freedom from TLR and reocclusion.7 Furthermore, Gandini et al demonstrated that TLR, patency, and amputation rates were significantly improved with laser + DCB treatment compared with DCB alone.8 Laser atherectomy helps prepare the vessel prior to DCB angioplasty by increasing the amount of exposed lumen for maximum drug delivery. Thus, to maximize treatment effectiveness and durability, all patients included in this analysis received adjunctive therapy at the time of the procedure consisting of balloon angioplasty, stent placement, or both.
Previous published literature has demonstrated that laser atherectomy can safely and effectively treat SFA lesions as a useful vessel-preparation tool.9 However, evidence to guide optimal device setting is scant. Each setting is associated with a different photomechanical profile. The absorption of light, frequency of acoustic pressure wave, and size of the cavitation bubble varies with the specific device settings. We hypothesized that these factors would have different effects on luminal gain depending on lesion morphology.
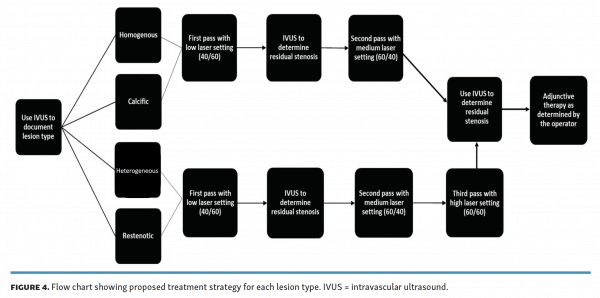
The results of our study support this hypothesis. The homo- genous lesions saw minimal or no luminal gain after a third pass with the high laser setting, which implies that atherectomy in this type of patient can exclude high-intensity passes with no significant clinical impact. Heterogeneous and restenotic lesions saw significant gain in luminal area following the third pass, suggesting that all 3 sequential laser settings should be used when treating such lesions. Calcific lesions showed a benefit with advancement to the high-intensity setting; however, the overall luminal gain was reduced as compared with treatment of heterogeneous and restenotic lesions. Figure 4 compiles these findings into a flow chart that proposes an algorithmic approach that pairs lesion type to atherectomy setting and technique.
The material properties of the different lesion types interact with the photomechanical energy exerted by the laser atherectomy. For example, we might expect plaque with less calcium to absorb more energy and undergo more mechanical deformation after the first pass with the laser. In contrast, highly calcific plaque may not absorb the same amount of energy and thus is not deformed to the same extent. Additional ex vivo studies may be needed to validate this hypothesis.
IVUS has recently emerged as the premier adjunctive imaging/measurement modality for peripheral arterial interventions. In their 2019 publication, Shammas et al compared IVUS with angiography — the most widely utilized imaging modality during vascular procedures.10 Clinical outcomes obtained with IVUS-guided treatment were superior to those obtained with treatment guided by angiography only. The authors proposed that this was because IVUS can more precisely image vessel size and plaque morphology, as well as more precisely discern the presence of dissections. Furthermore, this imaging modality is a useful guide for assessing residual narrowing, stent sizing and apposition, and completeness of stent expansion during interventional procedures.10 Therefore, we propose that IVUS is an essential imaging tool to ensure accurate plaque characterization to ensure that the vessel is adequately prepared for all adjunctive procedures and that complications do not go unrecognized.
PAD is a diverse disease and tailoring treatments to the specific disease of the patient can improve the success and safety of treatments and help personalize care. This pilot study provides some preliminary insight into the optimal use of laser atherectomy to specific lesion morphology. There remains considerable opportunity for progress, and one area for future study could include correlation of luminal gain to clinical outcomes, and identification of minimal luminal gain, which is safe and clinically meaningful. In answering these questions, we can potentially streamline treatment procedures, optimize patient and physician time, and utilize resources more efficiently. In addition, defining the optimal strategy for pairing plaque morphology with laser atherectomy settings may have an impact on reducing procedure times and costs by eliminating extraneous passes with the laser catheter in morphology types that do not result in clinically meaningful luminal gain.
Study limitations
This was a pilot study conducted at a single center with a relatively small sample size. This could lead to unintended selection bias and other confounding factors. Additionally, follow-up was limited. While patient medical records were reviewed for adverse events up through 1 year, only the initial 30-day follow-up visit was mandated to be in person in this study. This could result in potential missed adverse events and inaccuracies in efficacy evaluations.
Conclusion
This pilot study evaluated different excimer laser settings during laser atherectomy of SFA and above-knee popliteal artery lesions, and found that luminal gain varied according to lesion type. Patients with heterogeneous and restenotic lesions achieved the greatest benefit from more aggressive laser therapy settings, whereas patients with homogenous and calcific lesions had little to no incremental improvement when a pass with high-intensity laser setting was performed after low and medium passes. IVUS is a particularly useful procedural tool and augments intraprocedural decisions about choice of plaque modification and treatment modalities by characterizing plaque morphology and accurately determining luminal gain. Further studies are needed to help develop validated interventional strategies for peripheral artery disease.
Acknowledgments
Jennifer Ferguson (NC Heart and Vascular Research, LLC) assisted in the collection and reconciliation of the data and images, with support on image acquisition from Hailey Austin and Jennifer Morin-Ebarb (Spectranetics/Philips). Kasthuri Nair (Syntactx) assisted in the writing and editing of this report. Priyanka Kumari and Chao Cheng (Syntactx) assisted with statistical and data analysis.
Disclosures
From the University of North Carolina, Chapel Hill, North Carolina; and 2UT Southwestern Medical Center, Dallas, Texas.
Disclosure: The authors have completed and returned the ICMJE Form for Disclosure of Potential Conflicts of Interest. Dr Adams reports grant support and personal fees from Philips. Dr Subramanian reports no conflicts of interest regarding the content herein.
Manuscript accepted February 12, 2021.
Address for correspondence: George L. Adams, MD, NC Heart and Vascular Research, 4414 Lake Boone Trail, Suite 409, Raleigh, NC 27607. Email: George.adams@unchealth.unc.edu
To download a PDF of this article visit CLIJournal.com